On Saturday, President Trump signed an executive order to accelerate the research, approval, and deployment of psychedelic medicines for serious mental illness. For those of us who have spent years pushing this field toward legitimacy, it’s a remarkable moment. A directive to the FDA and DEA. Federal money. VA partnerships. A Schedule I review. Breakthrough Therapy vouchers. Data-sharing mandates. And maybe most importantly, a Republican administration staking a clear public position in favor of psychedelic medicine. With bipartisan support. (I mean, what has bipartisan support these days?)
That’s huge.
But it’s also not the whole picture. If you only read the EO, you’d think the story of psilocybin and ibogaine is a story about treating diagnosed disease. It isn’t. Or more precisely: it’s that, and it’s a great deal more.
What the EO actually does
The order directs the FDA to issue National Priority Vouchers to psychedelic drugs already holding Breakthrough Therapy designation, to stand up Right-to-Try pathways for eligible patients, and to begin Schedule I reviews for compounds that complete Phase 3 trials. It commits at least $50 million through ARPA-H to support state programs. It brings the VA into clinical-trial partnerships and data-sharing, which is long overdue given that veteran suicide rates remain more than twice the non-veteran rate, and have for two decades.
The scope is “serious mental illness”: diagnosable conditions that substantially interfere with daily functioning. Major depressive disorder. Substance use disorder. PTSD. The roughly 14 million American adults living with SMI today. This matters enormously. These are people whose suffering is both immense and, in many cases, treatment-resistant. Anything that accelerates access to therapies that actually work for them is a moral good.
Why it’s only part of the story
The EO’s framing, and the FDA approval pathway it accelerates, is bound by diagnosis. This makes sense, it’s how the regulatory machinery works. A drug gets approved for a specific indication. Clinicians prescribe within the bounds of that indication. Insurance reimburses for that indication. The entire infrastructure of American medicine is built on the diagnosis-treatment pairing. Sick care, as it’s affectionately referred to by its critics.
Psychedelics don’t fit that shape particularly well.
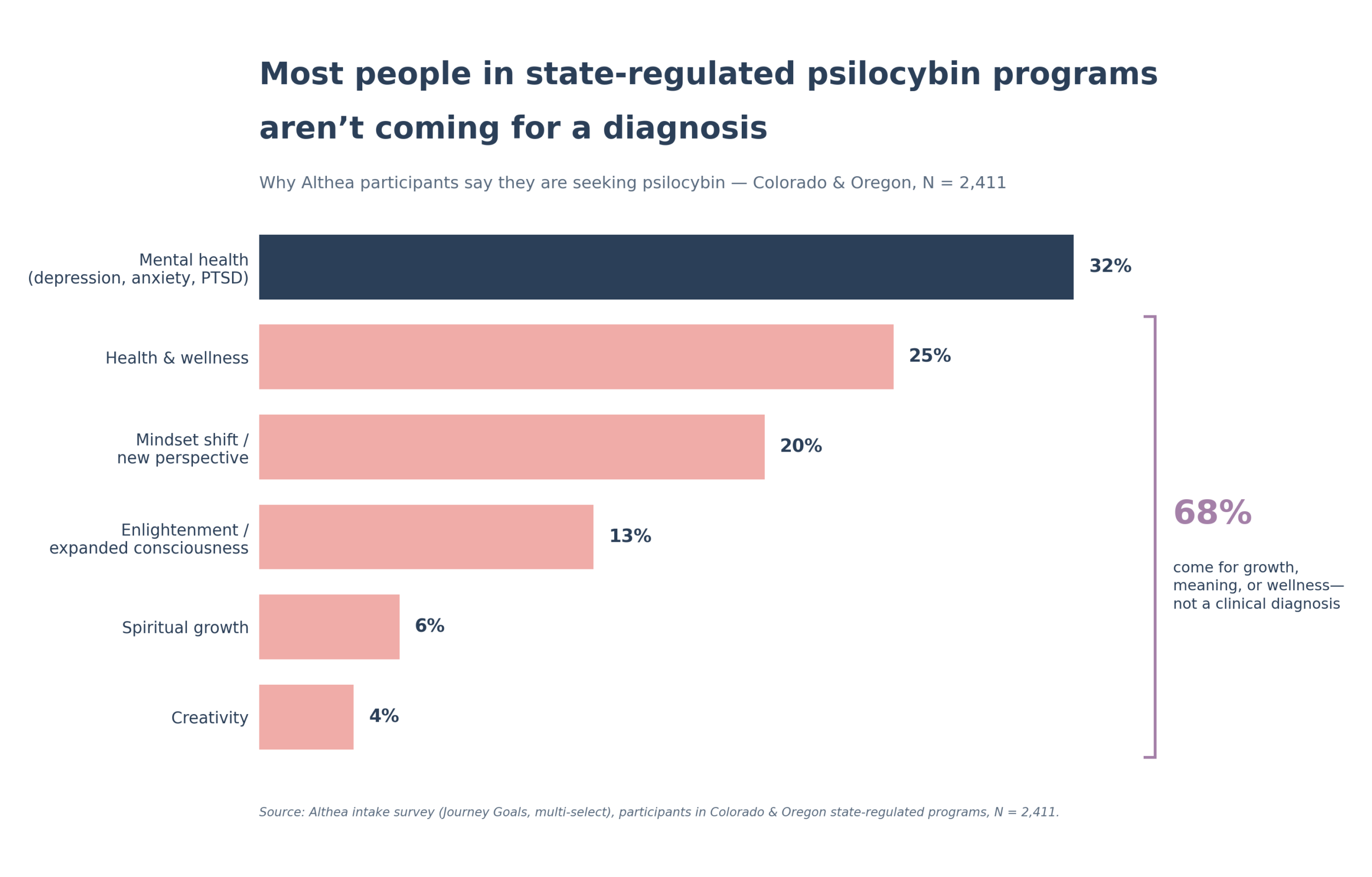
At Althea, we support the operations of psychedelic centers in Colorado and Oregon’s state-regulated psilocybin programs. By design, these programs do not require a diagnosis. People come to us for a lot of reasons. Some of them are wrestling with depression or anxiety or PTSD. Most are not. Our intake data from more than 2,400 participants makes this striking:
Most people in state-regulated psilocybin programs aren’t coming for a diagnosis
Why Althea participants say they are seeking psilocybin. Colorado & Oregon, N = 2,411.
(depression, anxiety, PTSD)
new perspective
expanded consciousness
Source: Althea intake survey (Journey Goals), participants in Colorado & Oregon state-regulated psilocybin programs, N = 2,411. Percentages reflect the share of participants selecting each primary reason for seeking psilocybin.
About 32% of participants name a mental-health concern as their reason for coming. The other 68% are there for wellness, for a change of perspective, for spiritual growth, for what they describe as expanded consciousness, for creativity. They are grieving. They are trying to become better parents, better partners, better humans. They are at the edge of something in their lives (a marriage, a career, a loss, a decade), and they want help moving through it.
This is not a population the FDA approval pathway was ever designed to reach. Not because these people don’t deserve care, but because what they’re seeking isn’t a treatment for a disease. It’s something older and broader: a kind of medicine that the cultures who used these plants for centuries understood as belonging to the category of meaning-making, not medication.
A rising tide, not a zero-sum choice
Some in the field have worried that a federal embrace of the medical model will undercut state-regulated programs. I think the opposite is true. The EO is a rising tide.
The biggest thing holding back state programs isn’t regulation, supply, or training. It’s stigma. It’s the lingering sense among reasonable, thoughtful Americans that psychedelics belong to the counterculture, something you do at a festival, not something you consider to move through your mother’s death, or a divorce, or a crisis of meaning in middle age. A Republican administration, an FDA fast-track, a Schedule I review, $50M through ARPA-H, RFK Jr., and Rogan on the same side of this issue has done so much to erode that stigma.
When the FDA approves psilocybin for depression, a skeptical relative won’t suddenly sign up. But their doctor’s willingness to mention it might shift. Their pastor’s willingness not to condemn it might shift. The range of Americans for whom this becomes thinkable expands dramatically.
That cultural permission flows downstream to the state programs too. Every FDA approval normalizes the molecule. Every veteran who finds relief through VA-sponsored research rewrites the public narrative. Every insurance code and every CME course builds familiarity. All of that makes it easier, not harder, for someone to walk into a Colorado or Oregon facilitator’s office for reasons that aren’t a diagnosis at all.
Two pathways, one direction
The clinical pathway will open access for the people who need a diagnosis to justify the spend, a prescription to convince a spouse, an FDA label to persuade their doctor or payer. The state-regulated pathway is already serving a much broader population, one the medical model was never built to reach, and one that will grow tremendously because of medical adoption.
Both pathways are necessary. Both are mutually validating. And as of Saturday, both just got a very big wind at their back.